VA SOL's
PS.3: The student will investigate and understand the modern and historical models of atomic structure. Key concepts include
a) the contributions of Dalton, Thomson, Rutherford, and Bohr in understanding the atom; and
b) the modern model of atomic structure.
PS.4: The student will investigate and understand the organization and use of the periodic table of elements to obtain information. Key concepts include
a) symbols, atomic numbers, atomic mass, chemical families (groups), and periods;
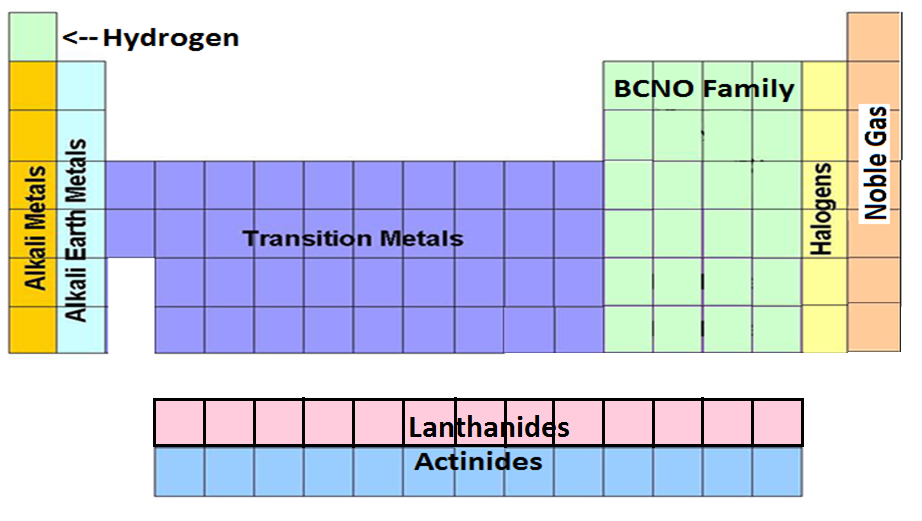
b) classification of elements as metals, metalloids, and nonmetals; and
PS.5: The student will investigate and understand changes in matter and the relationship of these changes to the Law of Conservation of Matter and Energy. Key concepts include
c) nuclear reactions.
PS.11: The student will investigate and understand basic principles of electricity and magnetism. Key concepts include
d) conductors, semiconductors, and insulators.
a) the contributions of Dalton, Thomson, Rutherford, and Bohr in understanding the atom; and
b) the modern model of atomic structure.
PS.4: The student will investigate and understand the organization and use of the periodic table of elements to obtain information. Key concepts include
a) symbols, atomic numbers, atomic mass, chemical families (groups), and periods;
b) classification of elements as metals, metalloids, and nonmetals; and
PS.5: The student will investigate and understand changes in matter and the relationship of these changes to the Law of Conservation of Matter and Energy. Key concepts include
c) nuclear reactions.
PS.11: The student will investigate and understand basic principles of electricity and magnetism. Key concepts include
d) conductors, semiconductors, and insulators.
Objectives
- I can describe how scientists have contributed to our modern understanding of the atom.
- I can differentiate among the three basic particles in the atom (proton, neutron, and electron).
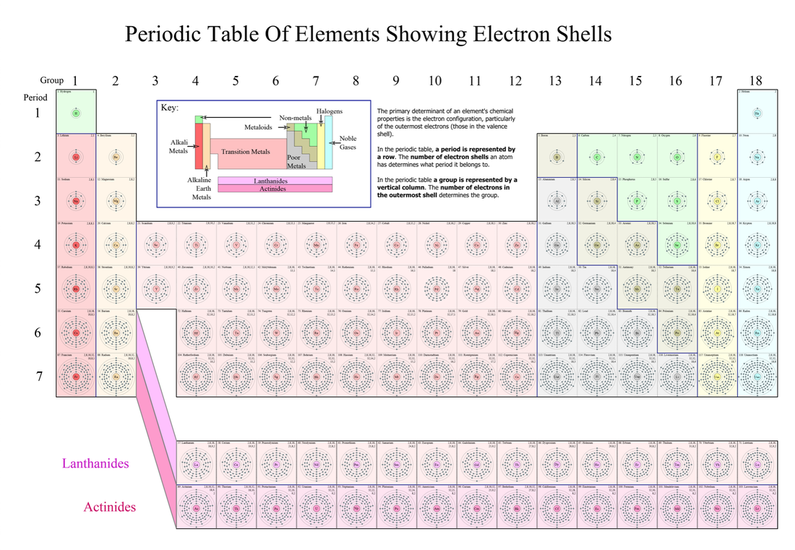
- I can use the Periodic Table to determine what an element's atoms might look like.
- I can use the Periodic Table as a tool to determine properties about the elements.
- I can determine if bonds between atoms are ionic or covalent.
- I can use the pH of a substance to determine if it is an acid, base, or neutral.
- I can demonstrate the Law of Conservation of Mass by balancing chemical equations.
Key Terms
|
|
Essential Questions
Can you answers these questions? By the end of this unit you should be able to!!
Element Symbols to Memorize
|
|
|
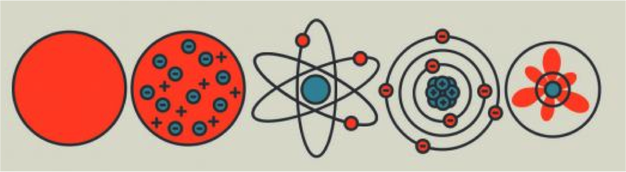
History of the Atom
|
|
|
|
|
|
|
|
|
What Do We Know About Atoms?
(parts of the atom, reading "periodic squares," Bohr models)
|
|
|
|
|
|
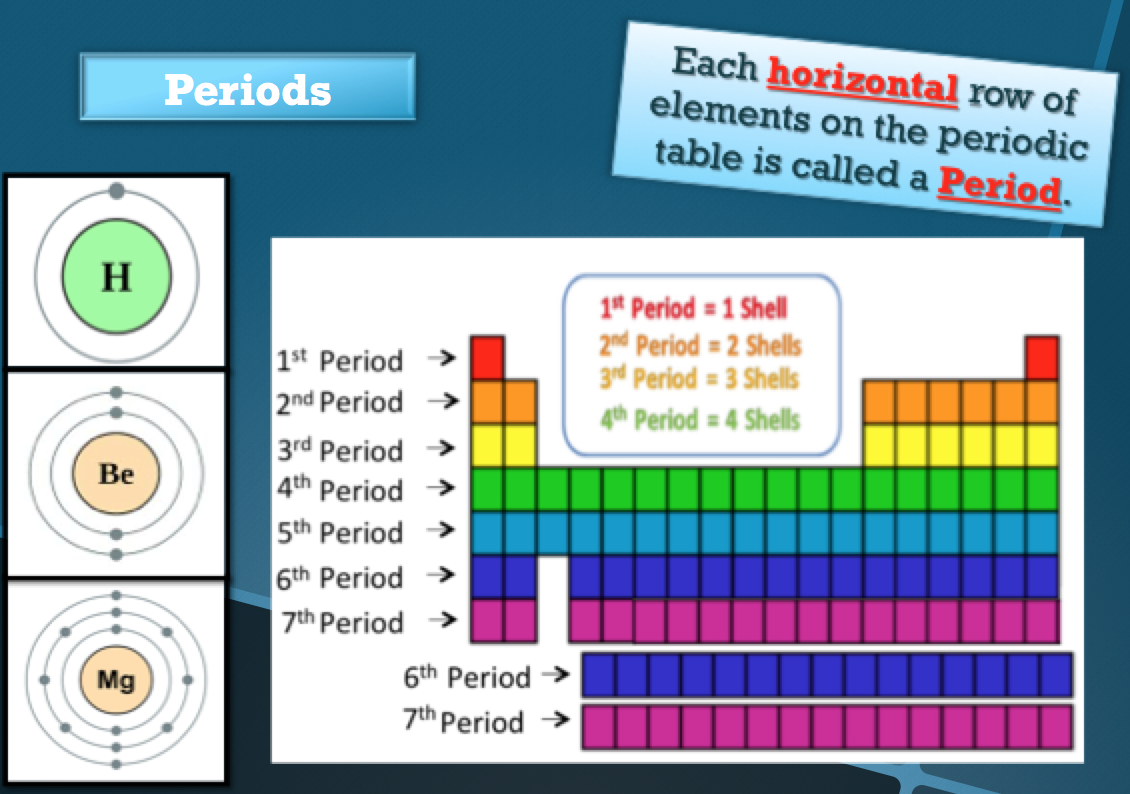
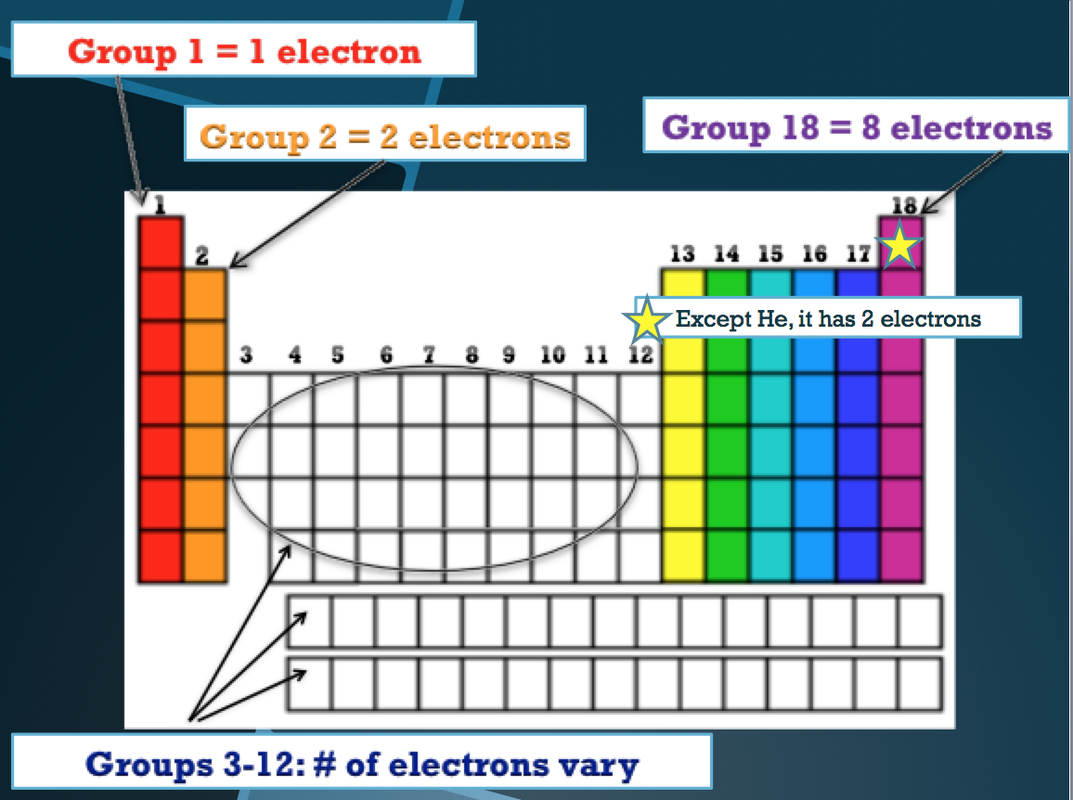
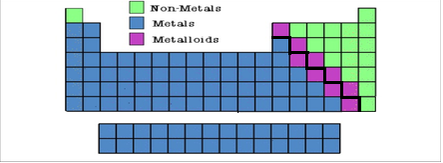
How is the Periodic Table Organized?
Periods, Groups, Families, Classes
|
|
|
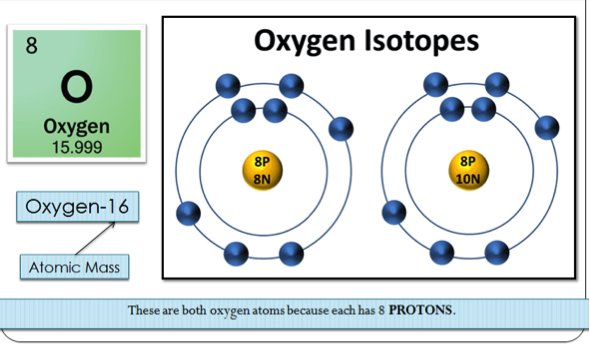
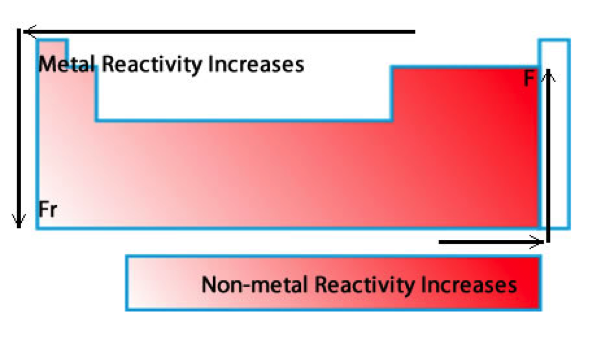
Isotopes & Reactivity
How do atoms bond together?
|
Ionic Bonds
|
Covalent Bonds
|